Multiply this by about 0.75 to account for real cycle effects (such as the time it takes to burn, heat losses to the coolant, and exhaust valves that open before the piston fully reaches bottom position) and you have h = 0.35. Plug g = 1.27 into the efficiency equation above, assume r v = 10, and you get h = 0.46. Thus, its g is higher, and its h is greater. A lean engine (ie, an engine with excess air) has a cooler combustion process and more air relative to fuel than the typical engine with a chemically correct mixture. Thus, as g drops (indicating more vibration of the molecules), h drops. The other modes of molecular motion do nothing for pushing the piston. However, only the translation of the molecules PUSHES the piston. Molecules of fuel vapor have a lot of opportunity to vibrate, even at room temperature. Hot air starts to vibrate (as two nuclei connected by a spring). Room air molecules translate and rotate (about 2 of their axes). Argon and helium atoms only translate, that is, they move along straight paths until they encounter another atom. The rule of thumb is: the greater the complexity of the molecules, the lower the g. The overall, effective g for the whole cycle for use in the efficiency equation above is about 1.27. Upon combustion (when the piston is near its top position), the fuel is oxidized to CO 2 (and some CO) and H 2O, and g drops further. As this mixture is compressed and heated during the compression stroke, its g drops to about 1.33. The mixture of air and gasoline vapor inducted into the engine has a g of about 1.35. Room air on the other hand, being mainly composed of O 2 and N 2 molecules has a g of 1.4. A gas such as helium or argon, composed only of atoms, has the highest g possible, 1.67. In practical terms, the higher the g, the higher the efficiency. 
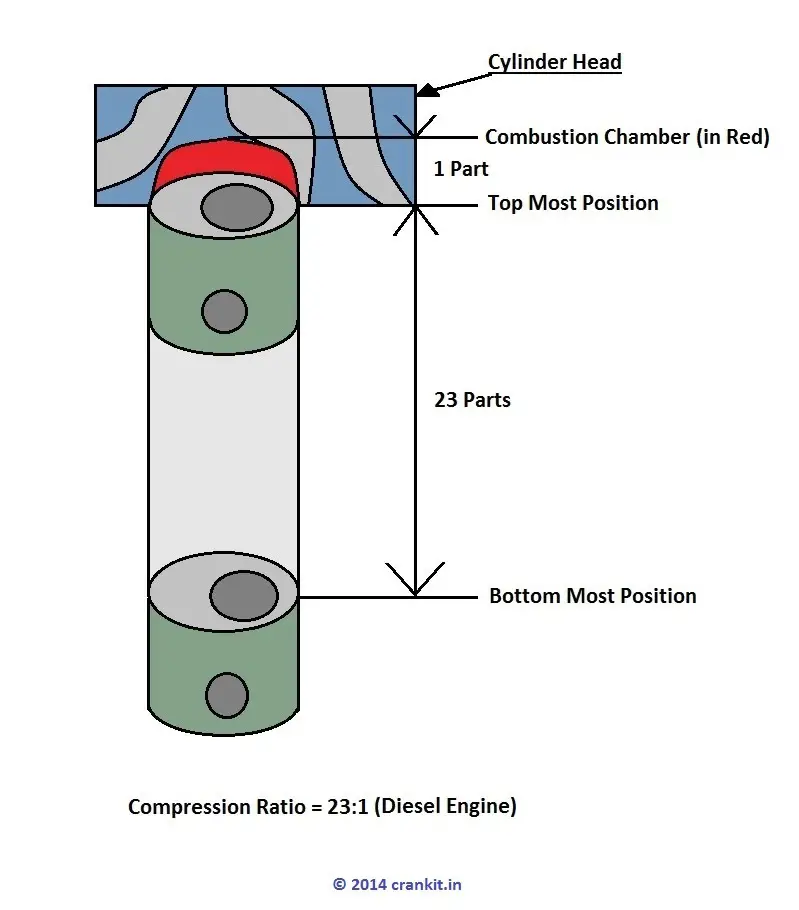
We note: the higher the compression ratio, the higher the efficiency! The g parameter is the ratio of the specific heats, ie, the constant pressure specific heat over the constant volume specific heat. Most auto engines have compression ratios in the 9 to 10.5 range. It is the ratio of the volume in a cylinder when the piston is at the bottom of the cylinder to the volume in the cylinder when the piston is at its top position: r v = V bottom/V top. The compression ratio of the engine is r v. Such a course would derive the following equation for the Otto cycle efficiency: If the gasoline-air mixture is too lean, the flame will not have enough speed to get across the cylinder in the time permitted by the engine RPM the driver wants, or the flame will not even start – the cylinder misfires, and then the catalyst has to oxidize a huge amount of UHC and thus may overheat (which might mean you have to buy a new catalyst).Ī first course on thermodynamics may teach the efficiency of the Otto cycle (which is the ideal cycle used to simulate the gasoline spark ignition auto engine). That is, the ability to burn real lean is limited by the fuel. Then we could have increased efficiency and continue to be clean!Īlso needed are ways to improve lean flammability in gasoline engines. (UHC = unburned hydrocarbons.) What humankind needs is a lean-NOx catalyst. Only at this ratio can the catalyst both oxidize the CO and UHC (to CO 2 and H 2O) and chemically reduce the NOx (to N 2). The engine computer, acting in concert with the engine air flow sensor, electronic fuel injectors, and exhaust oxygen sensor, maintains the stoichiometric ratio for most of your driving. This only works if the engine air/fuel ratio (by mass) is stoichiometric (chemically correct). So what happen to change this? The problem is the three-way (CO, UHC, NOx) catalyst used on engine exhausts. In the old days, under cruising conditions, the engines always ran lean – about 15% excess air - this was economical. It is well known that fuel-lean running improves the efficiency. Run the engine fuel-lean, that is, use excess air.OVERCOMING ENGINE FRICTION AND PUMPING THE AIR AND FUELĪre we stuck with ~20% auto engine efficiency? 
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
